Introduction
For this project we were given the main task of creating a board game that included chemical reactions. Our game had four reaction requirements that we had to have in the game. We had to have a single displacement reaction, a double displacement reaction, production of a gas, and lighting an LED. My group consisted of four people Ryan, Joell, Nitin, and myself. We contributed a different element to our groups project.
Each of us chose one of the requirements to come up with our own reactions. Personally I chose production of a gas. Due to the absence of one of our members for the majority of the project, we combined the LED and single reaction. We all collaborated and decided we wanted to base our board game around candyland. We created a blueprint and spent the next three weeks building and creating our project.
Each of us chose one of the requirements to come up with our own reactions. Personally I chose production of a gas. Due to the absence of one of our members for the majority of the project, we combined the LED and single reaction. We all collaborated and decided we wanted to base our board game around candyland. We created a blueprint and spent the next three weeks building and creating our project.
Evidence of Work

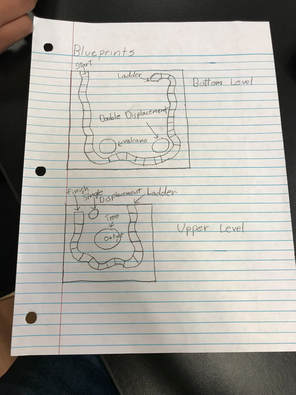
Our board game consisted of two boards stacked on top of one another. The spaces wrap around the board and end on the top. There are two reactions on the bottom board and one on the top.
How to Play:
1. On every turn, each player must pick a card from the pile and move their piece to that colored square
2. There are two reactions throughout the game and one to win, you must successfully complete the reactions to move on
3. The first player to light the led win
4. If you don´t create the reaction you must wait till your next turn to try again
How to Play:
1. On every turn, each player must pick a card from the pile and move their piece to that colored square
2. There are two reactions throughout the game and one to win, you must successfully complete the reactions to move on
3. The first player to light the led win
4. If you don´t create the reaction you must wait till your next turn to try again
Blueprint
Reactions
Double Displacement Reaction
Production of Gas
Single Displacement Reaction
- First we will prepare a solution of Lead Nitrate. For this we will pour about 1 mL of Lead Nitrate in an empty beaker.
- Then we pour some water into the Lead Nitrate beaker. This will be a saturated solution of Lead Nitrate.
- Similarly, we will also prepare of solution of Potassium Iodide. pour about 1 mL of Potassium Iodide in an empty beaker, add some water and stir to make a solution of Potassium Iodide.
- The Potassium Iodide solution will be transparent like water, while the Lead Nitrate solution will be milky in color.
- Now add the Lead Nitrate solution into the Potassium Iodide solution. Immediately a reaction will take place.
Production of Gas
- Place two spoonfuls of baking soda in the volcano
- Add 45 ml of vinegar to volcano
- Baking soda and vinegar react with each other because of an acid-base reaction. Baking soda is a bicarbonate (NaHCO3) and vinegar is an acetic acid (HCH3COO). One of the products this reaction creates is carbon dioxide.
Single Displacement Reaction
- Add blue solution to small petri dish
- Aluminum foil already added
Content
Single Displacement- An element reacts with a compound and takes the place of another element in that compound
Double Displacement- Chemical reaction where two compounds react, and the positive ions (cation) and the negative ions (anion) of the two reactants switch places, forming two new compounds or products
Synthesis- A purposeful execution of chemical reactions to obtain a product, or several products. This happens by physical and chemical manipulations usually involving one or more reactions.
Decomposition- Analysis or breakdown is the separation of a single chemical compound into its two or more elemental parts or to simpler compounds.
Combustion- Rapid chemical combination of a substance with oxygen, involving the production of heat and light.
Law of conservation of mass/matter- For any system closed to all transfers of matter and energy, the mass of the system must remain constant over time, as system's mass cannot change, so quantity cannot be added or removed, quantity of mass is conserved over time
Balancing chemical equations- Using one type of equation and having it go through a reaction, but come out with the same chemicals, in a different order on the other
Production of a gas- Two chemical that mix to form some type of gas
Double Displacement- Chemical reaction where two compounds react, and the positive ions (cation) and the negative ions (anion) of the two reactants switch places, forming two new compounds or products
Synthesis- A purposeful execution of chemical reactions to obtain a product, or several products. This happens by physical and chemical manipulations usually involving one or more reactions.
Decomposition- Analysis or breakdown is the separation of a single chemical compound into its two or more elemental parts or to simpler compounds.
Combustion- Rapid chemical combination of a substance with oxygen, involving the production of heat and light.
Law of conservation of mass/matter- For any system closed to all transfers of matter and energy, the mass of the system must remain constant over time, as system's mass cannot change, so quantity cannot be added or removed, quantity of mass is conserved over time
Balancing chemical equations- Using one type of equation and having it go through a reaction, but come out with the same chemicals, in a different order on the other
Production of a gas- Two chemical that mix to form some type of gas
Reflection
This project overall was fun and I learned a lot. During this project I learned how to create and execute a single and double replacement reaction. This allowed me to further my understanding of how chemical reactions happen. I also learned how to create a breadboard that can light and LED. We used the breadboard to include in one of our reactions and as a requirement.
Although the project was successful there was a couple things to improve upon. First, our group could have been more productive. We could have finished a lot more on our bored paying attention to detail. We also could improve on our planning and blueprint skills. In the beginning we could have spent more time planning out the whole project.
Although the project was successful there was a couple things to improve upon. First, our group could have been more productive. We could have finished a lot more on our bored paying attention to detail. We also could improve on our planning and blueprint skills. In the beginning we could have spent more time planning out the whole project.